Overview
What are friends for? Is stress really bad? Why did language evolve? These are some of the questions that first inspired me to want to learn more, and today they drive my research program. I take a comparative approach to understanding the adaptive function of the stress response as a mediator of life history tradeoffs. Broadly, my interests center around how environmental challenges (both social and ecological) shape primate physiology, and how variation in individual behavioral responses to such challenges impact evolutionary fitness. I use a combination of methodologies to carry out this work, including behavioral observation, fecal hormone collection and analysis, and vocalization analysis in two species of wild primates: geladas (Theropithecus gelada: Cercopithecine primates endemic to Ethiopia) and white-faced capuchins (Cebus capucinus, in Costa Rica).
Research Interests
Behavioral endocrinology, stress physiology, social relationships, female reproductive strategies, social network analysis, the evolution of social behavior and language, vocal grooming
Current Research
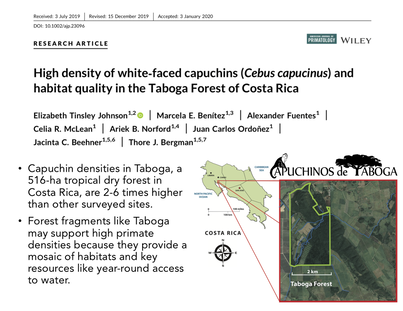 Research highlights from Tinsley Johnson et al., 2020
Research highlights from Tinsley Johnson et al., 2020
How does group size and composition interact with home range size and quality to impact fitness?
Female geladas in mid-sized units (4-7 adult females) have higher reproductive success than those in both small (<4 adult females) and large (>7 adult females) units (Tinsley Johnson et al., in prep). This is because large units experience more frequent alpha male replacements (i.e., takeovers), disruptive social events that negatively impact female reproduction via infanticide and the Bruce effect (Tinsley Johnson et al., 2018). Small units, on the other hand, experience higher rates of infanticide when takeovers do occur. Individuals are not necessarily “stuck” in less optimal units – instead, any given gelada will spend more time in a mid-sized unit than expected by chance. This is because large units tend to fission, and small units grow over time (and even fuse with other small units), a process that is mediated by the strength of female social ties. However, the number of adult females in mid-sized units exceeds the number of social partners a female gelada can maintain through grooming alone, raising the question of how these mid-sized units maintain social cohesion.
For female capuchins, group composition or habitat quality may matter more than group size. Our site in the Taboga Forest of Costa Rica contains an unusually high density of capuchins compared to nearby sites (Tinsley Johnson et al., 2020), meaning competition is high and intergroup encounters are frequent. I am interested in exploring how group size and composition relates to habitat quality, intergroup encounters, and fitness.
How do primates use vocalizations to mediate social relationships?
Geladas are exceptionally vocal primates, making them ideal for studying how vocalizations help manage social life (Gustison, Tinsley Johnson et al., 2019). My research demonstrates that a female gelada’s vocal contact network is actually larger than her grooming network, and vocal contact is primarily used when females are busy moving or feeding (Tinsley Johnson et al., 2014; Tinsley Johnson et al., in prep). Vocal contact networks may contribute to the cohesion of the social unit by allowing females to maintain larger social networks than they could through grooming alone, or may allow females to maintain critical social relationships even when time budgets are constrained at certain times of the year.
Capuchins use vocalizations in unique and flexible ways and have cultural traditions (e.g., tool use, unique social behaviors). Identifying between-group variation in vocal contact use can provide a better understanding of the evolution of ‘dialects’ in group dynamics. I am currently collecting data on the vocal repertoires of four groups at Taboga; specifically, I plan to look for variation in how combinations of certain vocalizations are used to efficiently mediate social interactions.
Female geladas in mid-sized units (4-7 adult females) have higher reproductive success than those in both small (<4 adult females) and large (>7 adult females) units (Tinsley Johnson et al., in prep). This is because large units experience more frequent alpha male replacements (i.e., takeovers), disruptive social events that negatively impact female reproduction via infanticide and the Bruce effect (Tinsley Johnson et al., 2018). Small units, on the other hand, experience higher rates of infanticide when takeovers do occur. Individuals are not necessarily “stuck” in less optimal units – instead, any given gelada will spend more time in a mid-sized unit than expected by chance. This is because large units tend to fission, and small units grow over time (and even fuse with other small units), a process that is mediated by the strength of female social ties. However, the number of adult females in mid-sized units exceeds the number of social partners a female gelada can maintain through grooming alone, raising the question of how these mid-sized units maintain social cohesion.
For female capuchins, group composition or habitat quality may matter more than group size. Our site in the Taboga Forest of Costa Rica contains an unusually high density of capuchins compared to nearby sites (Tinsley Johnson et al., 2020), meaning competition is high and intergroup encounters are frequent. I am interested in exploring how group size and composition relates to habitat quality, intergroup encounters, and fitness.
How do primates use vocalizations to mediate social relationships?
Geladas are exceptionally vocal primates, making them ideal for studying how vocalizations help manage social life (Gustison, Tinsley Johnson et al., 2019). My research demonstrates that a female gelada’s vocal contact network is actually larger than her grooming network, and vocal contact is primarily used when females are busy moving or feeding (Tinsley Johnson et al., 2014; Tinsley Johnson et al., in prep). Vocal contact networks may contribute to the cohesion of the social unit by allowing females to maintain larger social networks than they could through grooming alone, or may allow females to maintain critical social relationships even when time budgets are constrained at certain times of the year.
Capuchins use vocalizations in unique and flexible ways and have cultural traditions (e.g., tool use, unique social behaviors). Identifying between-group variation in vocal contact use can provide a better understanding of the evolution of ‘dialects’ in group dynamics. I am currently collecting data on the vocal repertoires of four groups at Taboga; specifically, I plan to look for variation in how combinations of certain vocalizations are used to efficiently mediate social interactions.
Past Research
My doctoral research focused on the costs and benefits of sociality, and the extent to which social relationships, and the behaviors that contribute to them, represent adaptive strategies that result in evolutionary fitness. Specifically, I studied the social relationships of female gelada monkeys (Theropithecus gelada) living in the Simien Mountains National Park, Ethiopia. I used a combination of methodologies to carry out this work, including behavioral observation, fecal hormone collection and analysis, vocalization analysis, and the use of the long-term University of Michigan Gelada Research Project dataset (including information on demography and genetics).
Geladas live in a highly seasonal afro-alpine ecosystem, where freezing temperatures and high altitude result in unusually high energetic demands. My research demonstrated that female geladas exhibit higher glucocorticoid levels and are less likely to conceive during the cold, wet season (Tinsley Johnson et al., 2018). These seasonal changes in thermoregulatory demands also shape the activity budget of individuals, requiring significant foraging time.
Increases in foraging time can cut deeply into social time. When time is limited, individuals must choose between focusing on a few close relationships, or spending time with each available partner, resulting in weaker ties. Although geladas forage in large bands (comprising hundreds of individuals), they actually keep their social networks small – that is, the actual geladas that they “know” is only a subset (Tinsley Johnson et al., 2014). This is in part because female geladas can only maintain 1-3 grooming relationships at a time. And, they are selective about who they socialize with, spending most of their social time with kin or with females of similar dominance rank.
Geladas live in a highly seasonal afro-alpine ecosystem, where freezing temperatures and high altitude result in unusually high energetic demands. My research demonstrated that female geladas exhibit higher glucocorticoid levels and are less likely to conceive during the cold, wet season (Tinsley Johnson et al., 2018). These seasonal changes in thermoregulatory demands also shape the activity budget of individuals, requiring significant foraging time.
Increases in foraging time can cut deeply into social time. When time is limited, individuals must choose between focusing on a few close relationships, or spending time with each available partner, resulting in weaker ties. Although geladas forage in large bands (comprising hundreds of individuals), they actually keep their social networks small – that is, the actual geladas that they “know” is only a subset (Tinsley Johnson et al., 2014). This is in part because female geladas can only maintain 1-3 grooming relationships at a time. And, they are selective about who they socialize with, spending most of their social time with kin or with females of similar dominance rank.
Vertical Divider
Funding
My research has been funded by the National Science Foundation (NSF-1340911, NSF-GRFP), the Leakey Foundation, and the University of Michigan.
Vertical Divider











